
Minoxidil Side Effects in 2026: What the Real Data Says
📌 TL;DR
- Most reported 'minoxidil side effects' on forums are either the dread shed (the drug working), propylene glycol contact irritation (formulation problem, not the drug), or finasteride side effects mis-attributed when people stack the two.
- Real topical side effects: scalp irritation ~6% (liquid only, PG-driven), unwanted facial hair <5% in women, dread shed at weeks 2–8 in most new users. No robust link to sexual dysfunction.
- Real oral LDOM side effects from 1,404-patient Vañó-Galván 2021: hypertrichosis 15.1%, edema 1.3%, palpitations 0.9%. Total discontinuation 1.7%.
- Cat toxicity is the only minoxidil side effect that is reliably fatal — to your cat, not you. Tiny amounts kill. Wash hands, dry fully before bed, store bottle out of reach. Non-negotiable if you live with cats.
- Pregnancy: do not use minoxidil. FDA category C, excreted in breast milk per LactMed. Both routes off-limits while pregnant or nursing.
Minoxidil Side Effects in 2026: What the Real Data Says
Last updated: May 2026 | Written by RK
Most of what’s online about minoxidil side effects falls into one of three buckets: the dread shed (the drug working, not failing), propylene glycol contact dermatitis (the formulation’s fault, not the drug), and finasteride side effects mis-attributed when someone is on both. The actual side-effect profile — the one that shows up in randomized trials — is narrower than the forum panic suggests, with a small number of real concerns that are worth taking seriously.
This is the deep-dive companion to the main minoxidil guide and the LDOM dosing & safety article. It separates real from imagined, and tells you what to do when something actually happens.

How to read the rest of this article
There are four tiers of “minoxidil side effect”:
The rest of this article walks each one in detail with the trial numbers, not the forum hearsay.
The dread shed — what’s actually happening to your follicles
The most common reason new users quit minoxidil at month 2 is the dread shed. The mechanism is well-characterized [1].

The shed happens on a calendar, not a clinical scale. Months 2–4 is when people quit at the worst possible moment.
Well-characterized in multiple RCTsWhat it is not: permanent loss. The hairs you shed during weeks 2–8 are coming out of follicles that have already started growing replacement hair underneath. The shed looks dramatic in the shower because shedding events are normally distributed across months — minoxidil compresses them into a few weeks.
What to do: nothing. Track photos under consistent lighting at week 0, week 8, week 16, week 24. If you’re still net-shedding past month 5, that’s worth raising with a dermatologist — at that point we’re talking about non-response, not dread shed.
Scalp irritation, itching, and propylene glycol contact dermatitis
This is the second-most common reason people quit. The Olsen 2002 randomized trial of 5% vs 2% vs placebo in 393 men documented application-site reactions in 7% of the 5% liquid group versus 4% in placebo [2]. The relevant comparator is what was in the liquid carrier, not the active drug.
The liquid solution uses propylene glycol (PG) as the primary solvent. PG dissolves minoxidil and pushes it through the stratum corneum effectively, but it’s also a well-known cause of allergic contact dermatitis when applied chronically to the scalp [3].
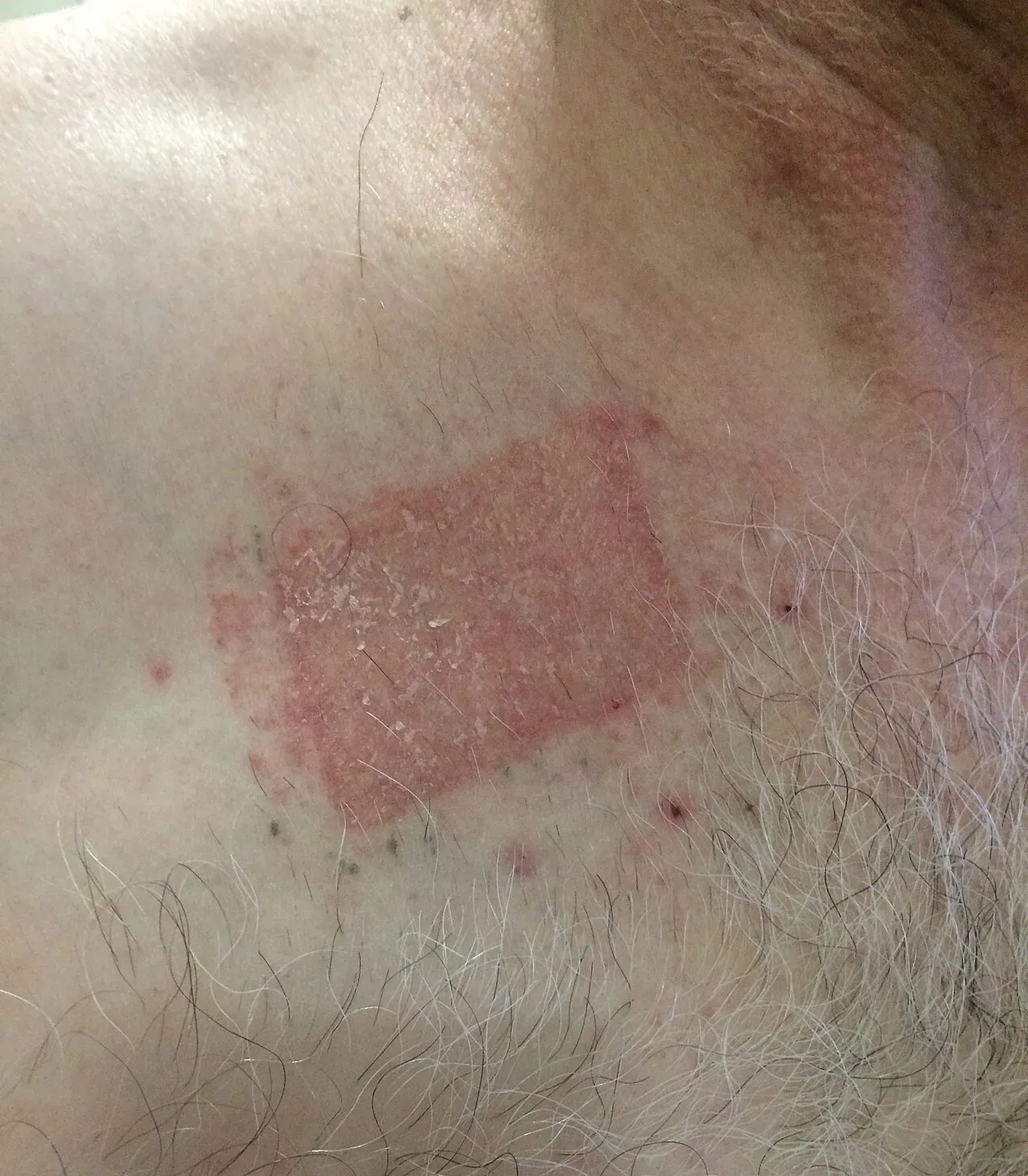
Severe contact dermatitis from a buprenorphine transdermal patch. Photo by Dr. khatmando, Wikimedia Commons, CC BY-SA 4.0. The same irritant pathway underlies PG-related minoxidil reactions on the scalp — itching, redness, scaling on the application zone.
What it looks like: red, itchy, flaky scalp localized to where you apply the drug. Sometimes the hairline forms a sharp boundary. Usually starts 1–4 weeks into use.
What to do, in order of cost:
1 Switch to foam. Same 5% drug, ethanol/cetyl alcohol instead of PG. Resolves the irritation in the majority of cases within 2–4 weeks.
2 If foam still irritates, try a compounded PG-free liquid. Some hair clinics compound minoxidil in glycerin or hydroxypropyl cellulose bases.
3 Add a 2× weekly ketoconazole wash to reduce baseline scalp inflammation. See the ketoconazole article.
4 If irritation persists across all topical routes, the cleanest move is to switch to oral LDOM (no scalp contact). See the LDOM guide.
Hypertrichosis — unwanted body and facial hair
Minoxidil is a hair-growth drug. It doesn’t know the difference between your scalp and your cheekbone. The rates differ sharply by route and sex.
How to manage it:
- Reversible: stop the drug, hair returns to baseline in 1–3 months.
- Dose reduction: usually proportional reduction in unwanted hair within 4–6 weeks.
- Cosmetic management: laser hair removal works on minoxidil-stimulated hair as well as on baseline hair.
- Switch to topical: if you’re on LDOM mainly for convenience, going back to topical drops the systemic exposure that drives facial hair growth.
For women specifically, the decision to start LDOM should include an honest conversation about the ~15% chance of needing routine upper-lip waxing or laser. Many find this an acceptable trade for the hair regrowth; some don’t.
Cardiovascular effects (oral minoxidil specifically)
This is where the dose makes the drug. At antihypertensive doses (10–40 mg/day, the original Loniten indication), minoxidil reliably causes reflex tachycardia and fluid retention serious enough to require concurrent beta-blocker and diuretic therapy. Pericardial effusion was reported and put a black-box warning on the label [7].
At LDOM doses (0.25–5 mg/day), this profile shifts dramatically. From the Vañó-Galván 2021 multicenter cohort of 1,404 patients:
Multicenter retrospective, n=1,404 (largest LDOM safety dataset)Total cohort discontinuation rate for adverse events: 1.7%. No hospitalizations were reported. The same study covered 7 clinics across 6 countries and a mean 7.7-month follow-up — the strongest case yet that LDOM in the 0.25–5 mg/day range is a different drug from Loniten in safety terms.
Where the trade still requires caution: patients on antihypertensives, beta-blockers, PDE5 inhibitors (Viagra/Cialis), or with unstable cardiac disease. Those scenarios need real physician monitoring, not telehealth checkboxes.
Topical minoxidil produces enough systemic absorption to detect in the bloodstream, but in healthy users it’s a fraction of LDOM levels and has not been associated with these effects in any meaningful frequency.
Cats — the one fatal side effect
This is the only minoxidil side effect that reliably kills, and it doesn’t kill you — it kills your cat. The mechanism is severe pleural effusion and cardiac failure from minoxidil’s vasodilatory action, to which cats are exquisitely sensitive even at trace doses [8].

🚨 The cat rule (non-negotiable)
Cats can die within hours of licking minoxidil residue. Documented routes include: licking your scalp, licking a contaminated pillowcase, knocking over a bottle and licking the spill, and licking the inside of a foam can. If you live with cats: wash hands and dry hair fully before bed, store the bottle in a closed cabinet, never leave foam cans out, and if you can’t guarantee these routines, switch to oral LDOM — no surface contamination to lick.
Pregnancy and breastfeeding
Both routes are off-limits.
Topical minoxidil: FDA pregnancy category C. Some systemic absorption occurs; animal teratogenicity data exist at high doses; insufficient human data to declare safe in pregnancy.
Oral minoxidil: clearly absorbed systemically; LactMed entry confirms transfer into breast milk [9]. Effects on a nursing infant are not well-studied but the precautionary default is to avoid.
If you’re already on minoxidil and become pregnant, stop the drug and consult your obstetrician. The hair you’ve gained will gradually revert to your baseline state over the following 3–6 months. That’s the trade.
What’s not a minoxidil side effect
A few persistent forum claims that don’t hold up against the trial data:
Erectile dysfunction / libido changes
Not documented for topical at any meaningful frequency. The almost-universal source of confusion: someone starts finasteride + minoxidil at the same time, develops finasteride-related sexual side effects, and attributes them to the drug they applied that morning. Stop finasteride alone for 4 weeks if you want to differentiate.
Hair turning gray faster
Not a documented effect. The mistaken observation: new hairs grown under minoxidil sometimes come in lighter or finer at first before darkening as they mature — and people interpret that as “graying.”
Permanent damage from stopping
Stopping reverses gains within 3–6 months back to where you would have been without treatment. That feels like “damage” but it isn’t. Your follicles haven’t been harmed — they’ve just lost the prop the drug was providing.
”Wrinkles” or premature aging of facial skin
Anecdotal claims circulate online; no controlled evidence supports them. The plausible confound is that PG-induced dermatitis on the forehead can produce dryness or scaling that some users attribute to aging.
When to actually stop
Most side effects are manageable through dose adjustment, formulation switch, or route switch. The shortlist for stopping outright:
You become pregnant
Stop both topical and oral immediately. Consult your OB.
Significant edema, persistent palpitations, or chest pain on LDOM
Stop. Recheck BP/HR. See a physician — these are dose-related and may require LDOM cessation, not just titration down.
Cat in the household and inability to maintain the safety routine
Stop topical. Switch to oral or stop entirely. This is not optional.
PG dermatitis that doesn’t resolve after foam switch + 4 weeks
Likely sensitivity to something beyond PG. Switch to oral LDOM or consider exiting minoxidil entirely.
For anything else — dread shed, mild facial fuzz on a woman, transient lightheadedness in the first week of LDOM — the right move is usually to wait, not to quit.
What to read next
- Minoxidil for Hair Loss: The Complete Guide (2026) — the pillar article. The full mechanism and routine if you’re new.
- Low-Dose Oral Minoxidil (LDOM) Guide (2026) — the dosing ladder, telehealth options, and the deep safety review.
- Finasteride Side Effects: What the Real Data Says (2026) — read this if you’re on both drugs and trying to attribute a side effect.
- Ketoconazole Shampoo for Hair Loss (2026) — useful as an anti-inflammatory adjunct if your scalp is reactive.
References
[7] FDA. “Loniten (minoxidil) tablet — full prescribing information.” U.S. Food & Drug Administration.
[8] Camp BJ. “Toxicology of minoxidil in domestic cats.” Vet Hum Toxicol. 1993;35(4):359-360.
[9] LactMed. “Minoxidil.” Drugs and Lactation Database (LactMed), National Library of Medicine.
Disclaimer: This article summarizes published evidence and is not medical advice. If you’re experiencing significant cardiovascular symptoms, dermatologic reactions, or pregnancy considerations, consult a licensed physician. The cat-toxicity warning is not a precaution — it is a hard rule. Topical minoxidil has killed cats in well-documented cases.


