
Minoxidil for Hair Loss: The Complete Guide (2026)
📌 TL;DR
- Minoxidil is the only FDA-approved over-the-counter topical for androgenetic alopecia. 5% beats 2% by ~46% in hair count (Olsen 2002).
- Foam (no propylene glycol) drops irritation rates significantly vs liquid — the practical difference for adherence is huge.
- Low-dose oral minoxidil (0.25–5 mg/day) is the fastest-growing prescription in dermatology — works systemically, no scalp application needed.
- Initial shedding at weeks 2–8 is normal — the drug pushes resting follicles into a new growth phase. Don't quit during the dread shed.
- Stopping minoxidil reverses gains within 3–6 months. It's a maintenance drug, not a cure.
Minoxidil for Hair Loss: The Complete Guide (2026)
Last updated: May 2026 | Written by RK
Minoxidil is the only over-the-counter topical the FDA has approved for androgenetic alopecia (AGA), and it’s the most-studied hair regrowth drug on the market. If you’ve been told to start treatment by three different sources, all of them mentioned minoxidil. Whether they agreed on which form, dose, or route is a different question.
This guide unpacks every decision: 5% vs 2%, foam vs liquid, topical vs oral, what to expect month-by-month, and when minoxidil isn’t actually the right tool.

What minoxidil is, and how it ended up in your bathroom
Minoxidil was developed by Upjohn (now part of Pfizer) in the 1950s as an oral antihypertensive — a blood pressure drug. It worked. It also had a side effect that the original researchers found inconvenient and the bald population found extremely interesting: patients on it grew thicker, darker hair across their bodies.
By the late 1970s, dermatologists were experimenting with topical formulations. In 1988, the FDA approved 2% topical minoxidil under the Rogaine brand for AGA in men [1]. The 5% formulation followed in 1991. Foam came in 2006. The drug went generic, the patent expired, and now the same active ingredient is sold under dozens of brand names — Rogaine, Kirkland, generic store-brands — all containing identical 2% or 5% minoxidil solutions.
In 2018, dermatologists started prescribing low-dose oral minoxidil off-label for AGA after a series of papers showed remarkable results at doses far below the original blood-pressure range [6][7]. Oral minoxidil is now one of the fastest-growing categories in hair-loss prescriptions, and the FDA’s clinical guidance has been gradually catching up.
How it actually works on the follicle
Most articles online hand-wave the mechanism. Here’s the version that actually answers questions like “why does it cause shedding at first” and “why does it stop working when I quit.”
Minoxidil itself is a prodrug — it has to be converted to minoxidil sulfate by sulfotransferase enzymes in the scalp before it does anything useful. People with low scalp sulfotransferase activity make up part of the “non-responder” population (estimated 30–50% of users) [3]. A few labs (e.g. Daniel Alain’s “Minoxidil Response Test”) now offer a $100–150 saliva or hair-bulb assay that estimates this in advance — useful if you want to know before committing six months, but not strictly necessary.
Once activated, minoxidil sulfate has three documented effects:
Vasodilation
Opens potassium channels on smooth-muscle cells around scalp blood vessels, increasing local microcirculation to the follicle.
Telogen shortening
Pushes follicles in the resting phase to exit telogen early and enter the new growth phase faster than they would naturally.
Anagen prolongation
Extends the active growth phase, allowing each hair to grow longer and thicker before the next reset.
What minoxidil does not do: it doesn’t address DHT. The hormonal driver of androgenetic alopecia is untouched. That’s why minoxidil alone often plateaus around year 1–2 — the drug is fighting telogen, but DHT keeps miniaturizing follicles in the background. This is also why the strongest protocols stack minoxidil with a DHT-blocker (finasteride, dutasteride, or saw palmetto). One puts a lid on the pot; the other lowers the heat.
The “dread shed” at weeks 2–8 is also explained by the mechanism: when minoxidil pushes a wave of resting follicles into early termination of telogen, the existing hairs in those follicles fall out before the new growth replaces them. So you see a temporary increase in shedding before regrowth becomes visible. People who don’t understand this quit the drug at the worst possible moment. (For the underlying biology — what telogen and exogen actually are, and why every hair-loss drug targets a phase transition — see The Hair Growth Cycle Explained (2026).)

5% vs 2% — why 5% is the rational default for men
The cleanest data on concentration comes from Olsen et al. 2002, which randomized 393 men with AGA into three arms (5% topical, 2% topical, placebo) for 48 weeks [2]:
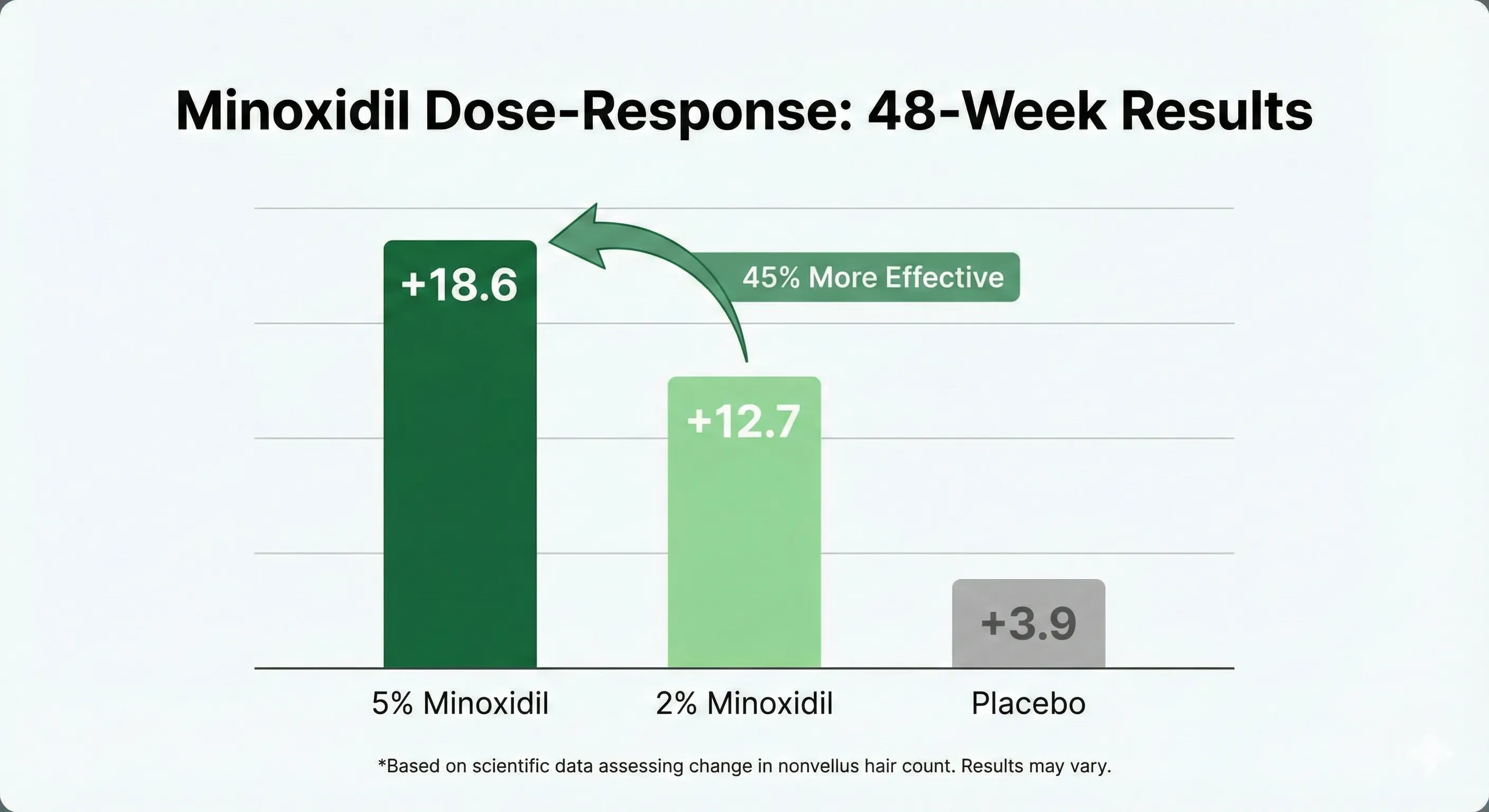
5% delivers 46% more hair count gain than 2%. There is no efficacy reason for a man to start at 2%, and the side-effect profile between concentrations is similar at typical doses.
For women, the FDA-approved dose is 2% — historically based on concerns about facial-hair side effects from systemic absorption. A separate Lucky 2004 trial in women showed 5% was modestly more effective than 2% with a slightly higher rate of unwanted facial hair growth [4]. In practice, many dermatologists now use 5% in women off-label and most tolerate it well.
Bottom line on strength: 5% if you’re male. 5% off-label or 2% on-label if you’re female. Don’t bother with anything below 2% — it’s commercial filler.
A 5% foam version exists too. Olsen 2007 tested it on 352 men and showed +21.0 hairs/cm² at 16 weeks, slightly outperforming the liquid version [5]. The likely explanation is better adherence — foam is much easier to apply daily.
Foam vs liquid — a decision driven by your scalp, not your wallet
The active ingredient is identical in both forms. The difference is the carrier solvent that delivers the drug into the scalp.
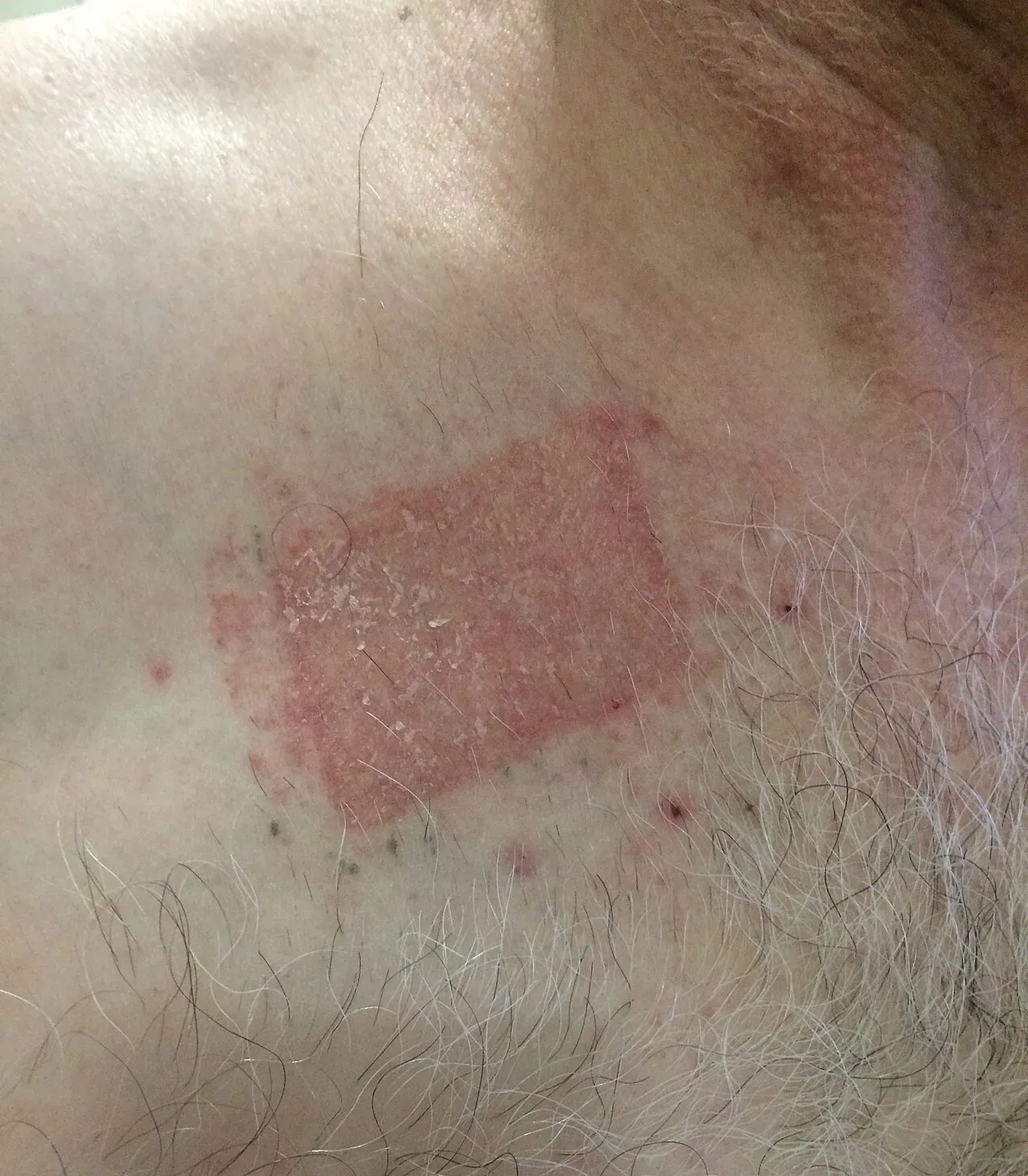
Severe contact dermatitis from a buprenorphine transdermal patch. Photo by Dr. khatmando, Wikimedia Commons, CC BY-SA 4.0. The same irritant pathway underlies PG-induced minoxidil reactions on the scalp.

The liquid solution uses propylene glycol (PG) as the primary solvent. PG is great at dissolving minoxidil and pushing it through the stratum corneum into the dermis, but it’s also the #1 cause of contact dermatitis on the scalp. Roughly 6% of minoxidil users develop PG-related itching, redness, or flaking [3].
The foam version drops PG entirely and uses ethanol/cetyl alcohol instead. Same active ingredient. Same dose. Much friendlier carrier.
For a deeper head-to-head with brand pricing, see Rogaine vs Kirkland Minoxidil (2026) — same active ingredient, dramatically different cost.
Topical vs oral minoxidil
Until 2018, “minoxidil” meant a topical solution applied twice daily. That assumption is now wrong.
Low-dose oral minoxidil (LDOM) at 0.25–5 mg/day, prescribed off-label by dermatologists, has become one of the fastest-growing categories in hair-loss prescriptions. The original blood-pressure dose was 10–40 mg/day; LDOM uses a small fraction of that.
What changed:
- A 2018 Sinclair paper documented strong efficacy at 0.25–1.25 mg/day in 100 women with FPHL [6]
- A 2021 JAAD review (Randolph & Tosti) of 17 studies and 634 patients confirmed LDOM as an effective and well-tolerated alternative for patients who can’t manage topical [9]
- A 2021 multicenter safety study (Vañó-Galván et al., n=1,404) showed a manageable side-effect profile across long-term use [7]
- A 2025 meta-analysis of randomized trials (Sobral et al.) found oral and topical minoxidil produced statistically equivalent improvements in hair density and diameter — meaning oral isn’t just a fallback, it’s clinically comparable to topical for AGA [10]
- Major dermatology centers now use LDOM as first-line for patients who can’t tolerate topical
Important: Oral minoxidil is prescription-only. It interacts with several common medications (especially other blood pressure drugs). Don’t source it without a physician — telehealth services like Hims, Roman, Keeps, and Happy Head all prescribe it after a consultation, but evaluation is required.
A more detailed deep-dive is on the way — see future article on the LDOM protocol when it’s published.
Side effects — the real numbers
Most worry about minoxidil is either inflated or specific to the liquid form’s PG content. The actual rates from RCT data are below — but read the cat warning first, because it’s the only one that’s actually fatal.

🚨 If you live with a cat: read this first
Topical minoxidil is fatal to cats — even tiny amounts. Cats groom themselves (and you) by licking. If your cat licks your scalp during the drying window, or licks your pillow, or licks the foam can, it can die within hours. This has happened to multiple owners who didn’t know. Wash hands thoroughly after every application, dry fully before bed, store the bottle out of paw reach. If you can’t guarantee this routine, oral minoxidil (no surface contamination) is a safer option in a cat household.
Scalp irritation, itching, flaking
~6% of liquid users. Almost entirely PG-driven. Switch to foam and most users resolve within 2–4 weeks.
”Dread shed”
Weeks 2–8. Affects the majority of new users to varying degrees. This is the drug working, not failing. Resolves by week 6 typically.
Unwanted facial hair (women, oral users of either sex)
Topical 5%: rare in women, mostly cheek/sideburn area. LDOM: more common (15–20%), reversible by stopping or dose reduction.
Cardiovascular (oral only)
Tachycardia, fluid retention, ankle swelling. Rare at LDOM doses (<5 mg/day) in healthy patients. Higher concern in patients with existing heart conditions — physician-monitored.
What’s not in the side-effect profile, despite frequent Reddit threads claiming otherwise: there is no robust evidence linking topical minoxidil to systemic libido or erectile dysfunction. That confusion typically comes from people stacking it with finasteride and attributing finasteride’s side effects to minoxidil.
What to expect month-by-month
The single biggest reason people quit minoxidil at month 3 is unrealistic expectations. Here’s the realistic timeline:
Weeks 1–2 Nothing visible. Drug is starting to enter follicle cycle.
Weeks 2–8 Dread shed possible. Hair counts may increase temporarily. Do not quit.
Months 2–4 Shedding stabilizes. Some users notice fine “vellus” hairs filling in thinning areas.
Months 4–6 First visible regrowth in most responders. Vellus hairs thicken into terminal hairs.
Months 6–12 Continued density gain. This is when before/after photos start showing meaningful difference.
Year 1+ Plateau. Most of the gain you’ll get is captured by month 12. Maintenance phase.
If you’ve been on minoxidil for 6 months at the right concentration, applied twice daily, with no visible response — you may be a low-sulfotransferase non-responder. Options include adding microneedling (boosts absorption), switching to oral, or stacking with a DHT-blocker.
How to apply (the part most people get wrong)

The pharmacology only matters if the drug actually reaches the dermis. Most users sabotage themselves on application. The protocol:
1 Apply on a dry scalp. Wet hair dilutes the drug.
2 Target the scalp, not the hair. The drug works on the follicle. Hair shafts don’t matter.
3 Liquid: 1 ml per application via dropper, 2× daily. Foam: half a cap, 2× daily.
4 Wait at least 4 hours before washing or sweating heavily. Penetration peaks around hour 1.
5 Wash hands afterward. Avoid contact with face, eyes, and anyone in the household if pregnant or a cat.
6 Skip a dose? Don’t double up. Just continue with the next scheduled application.
A common mistake worth flagging: people apply minoxidil right before bed and pull it onto a pillow, losing most of the dose. If you apply at night, give it 60+ minutes to dry on a liquid bottle, or use foam (10 minutes) for a more bedtime-friendly routine.

Combining minoxidil with other treatments
Minoxidil alone has a ceiling. The strongest AGA protocols use it as one ingredient in a stack:
The stack most dermatologists recommend for moderate AGA:
Finasteride 1 mg/day + Minoxidil 5% topical 2×/day + Microneedling 1.5 mm weekly
If you’re avoiding finasteride: replace it with saw palmetto 320 mg/day for a 50–60% efficacy approximation. See Saw Palmetto for Hair Loss in 2026 for the deeper write-up.
A microneedling-specific guide is on the way for the technique side.
When minoxidil isn’t the right tool
Minoxidil is broad-spectrum but not universal. Cases where it’s not the move:
Telogen effluvium (acute shedding)
If your loss is from a stress event, illness, postpartum, crash diet, or thyroid issue, minoxidil isn’t the right answer. The hair will grow back on its own once the trigger resolves. Treat the cause.
Alopecia areata (autoimmune patchy loss)
Different mechanism entirely. Treated with corticosteroids or JAK inhibitors. Minoxidil is not effective here.
Scarring alopecia
If the follicle is scarred and dead, no drug regrows it. Minoxidil only revives miniaturized but living follicles. Need a dermatologist diagnosis first.
Norwood 6–7 / very advanced AGA
Norwood 6–7 means extensive baldness with only a horseshoe of hair remaining. Past this point, follicles in fully bald areas are dormant or dead. Minoxidil can preserve the hair you have but won’t regrow large bald patches. Hair transplant becomes the realistic option.
The bottom-line decision tree
First-time user, mild loss, normal scalp
Kirkland 5% liquid, twice daily. Cheapest viable option, ~$5/month. Add foam later if scalp irritates.
Sensitive scalp or applying mornings
Kirkland 5% foam, twice daily. ~$10/month. Same drug, no PG, dries in 10 minutes.
Topical non-responder, or hate scalp application
LDOM, 1.25–2.5 mg/day. Requires telehealth (Hims, Roman, Keeps, Happy Head) or dermatologist visit.
Plateaued at 12 months on topical alone
Stack weekly microneedling (1.5 mm derma roller) and add a DHT blocker (finasteride or saw palmetto). Don’t just keep doing the same thing harder.
What to read next
- Rogaine vs Kirkland Minoxidil (2026) — once you’ve decided on minoxidil, this is the buy-side question: which brand and form, at what price?
- Saw Palmetto for Hair Loss (2026) — the natural DHT blocker that pairs cleanly with minoxidil if you want to avoid finasteride.
References
[1] FDA. “Minoxidil topical solution drug approval history.” U.S. Food & Drug Administration. 1988.
Disclaimer: This article is personal research and review. It is not medical advice. Before starting topical or oral minoxidil — particularly if you have cardiovascular conditions, low blood pressure, are pregnant, or live with cats — consult a licensed physician or dermatologist. Topical minoxidil is fatal to cats; this is a hard rule, not a precaution.


