Ketoconazole Shampoo for Hair Loss: What 27 Years of Evidence Actually Show (2026)
📌 TL;DR
- Ketoconazole 2% shampoo has 27 years of supportive (though imperfect) clinical data for androgenetic alopecia. Stronger evidence base than GHK-Cu, rosemary oil, or pumpkin seed oil — weaker than minoxidil or finasteride.
- Foundational trial — Piérard-Franchimont 1998 (n=39 men, vertex AGA): 27 men used 2% ketoconazole shampoo 2-4×/week vs 12 unmedicated. Hair density rose visibly by month 6, plateaued at month 15. Control group declined.
- El-Garf 2019 RCT (n=40 women, FPHL): 2% ketoconazole vs 2% minoxidil head-to-head. Both groups improved significantly at 6 months. Minoxidil acted faster (significant by month 4); ketoconazole caught up by month 6.
- Mechanism (Inui & Itami 2007): two pathways — (1) anti-androgenic activity (5α-reductase interference + steroidogenesis inhibition), (2) suppression of Malassezia yeast on the scalp, reducing inflammatory load.
- Fields 2020 systematic review (7 papers, 318 humans + 40 animals): conclusion is 'promising adjunctive therapy, more RCTs needed.' That's a measured take — not 'natural minoxidil replacement,' not 'doesn't work.'
- Practical use: 2% prescription Nizoral (where available) or 1% OTC. Apply 2-4×/week, lather, leave 3-5 minutes, rinse. Expect modest density and shaft-thickness improvement at 6 months. Cost: ~$10-25/month.
- Position: a legitimate adjunct to minoxidil and DHT blockers — particularly valuable because the active ingredient is in your shampoo (zero added routine time), it's cheap, and side effects are minimal.
Ketoconazole Shampoo for Hair Loss: What 27 Years of Evidence Actually Show (2026)
Last updated: May 2026 | Written by RK
If you’ve spent any time on hair-loss forums, you’ve seen the recommendation: use Nizoral 2-3 times a week, no exceptions, period. It’s been the default supplementary advice on r/tressless for over a decade. The recommendation is older than r/tressless — it’s older than Reddit. The first hair-density trial of ketoconazole shampoo was published in 1998.
Twenty-seven years of evidence later, the question worth asking honestly: what does that evidence actually show, and how should it weight relative to the other tools we have now?
Short answer: ketoconazole 2% shampoo is the best-evidenced over-the-counter topical for AGA other than minoxidil. The trials are imperfect, the effect is modest, but it’s been replicated across men and women, in vertex and frontal pattern, in mice and humans. It’s cheap, it slots into your existing routine for free, and the side-effect profile is benign. That’s a different category from a $90 GHK-Cu serum or unstudied “hair vitamin” gummies.
This article walks through every key trial, the actual mechanism, and where ketoconazole earns a place in a 2026 stack.
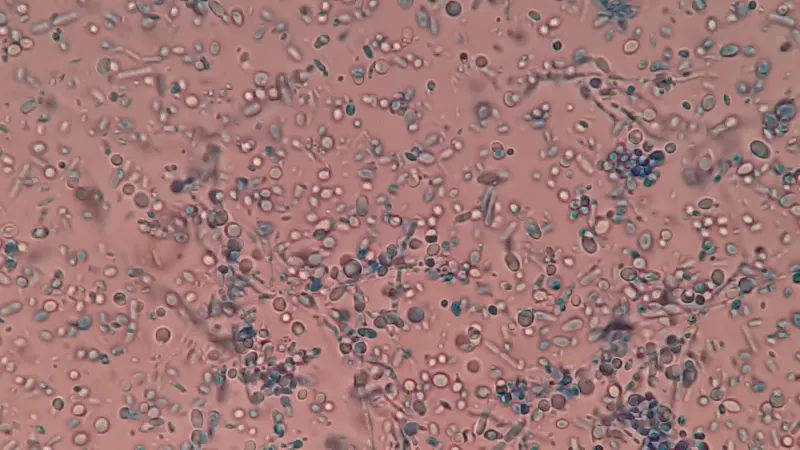
Photo: Ajay Kumar Chaurasiya via Wikimedia Commons (CC BY-SA 4.0). Malassezia yeast on the scalp is one of the targets ketoconazole acts on — though as we’ll see below, this isn’t the only mechanism that matters for hair loss.
TL;DR
- Best-evidenced OTC topical for AGA after minoxidil. 27 years of supportive trials, multiple replications.
- Foundational study (Piérard-Franchimont 1998): 39 men with vertex AGA, 21 months. Hair density rose visibly by month 6 in the ketoconazole arm, declined in unmedicated controls.
- Female-pattern replication (El-Garf 2019): 40 women, head-to-head with 2% minoxidil. Both worked. Minoxidil acted faster; ketoconazole caught up by month 6.
- Mechanism: dual — (1) anti-androgenic activity at the scalp (5α-reductase interference); (2) anti-Malassezia action reducing inflammatory load.
- Practical use: 2-4×/week, replacing your normal shampoo. 2% prescription strength was used in trials; 1% OTC is the practical default. Lather, sit 3-5 minutes, rinse.
- Cost: ~$10-25/month. Effectively free if you already shampoo.
- Position: legitimate adjunct to minoxidil and DHT blockers. Not a replacement for either.
The foundational trial: Piérard-Franchimont 1998
This is the trial that started everything. Published in Dermatology (Karger), authors Piérard-Franchimont, De Doncker, Cauwenbergh, and Piérard. The paper is actually two sub-studies in one publication:
Sub-study 1: ketoconazole vs unmedicated shampoo
- n = 39 men, ages 21-33, with grade III vertex androgenetic alopecia
- Exclusion: no diagnosed dandruff or seborrheic dermatitis in the prior 6 months (this matters — the hair benefit can’t be dismissed as “just treating dandruff”)
- Arms:
- 27 men: 2% ketoconazole shampoo, 2-4× per week
- 12 men: unmedicated shampoo
- Duration: 21 months
- Outcomes: hair density, hair shaft size, anagen-to-telogen ratio, sebum casual level
Result: hair density increased visibly in the ketoconazole group beginning around month 6, with continued rise through month 15, then plateauing. The unmedicated control group experienced declining hair density over the same period — i.e. AGA progression unchecked.
Sub-study 2: ketoconazole vs 2% minoxidil
- n = 8 men total (4 per arm), ages 24-29, grade III vertex AGA
- Arms: 2% ketoconazole shampoo (flexible frequency) vs 2% minoxidil daily
- Duration: 6 months
- Result: ~7% increase in hair shaft diameter in both groups
The honest methodological caveats:
- Not randomised in the modern sense. The arms used visibly different products (medicated shampoo vs unmedicated; shampoo vs liquid solution) — patients couldn’t be blinded.
- Sub-study 2 is tiny (n=4 per arm). The “comparable to minoxidil” claim that recurs in marketing has a very small evidence base.
- The unmedicated control was unfair. Real-world comparison would be against an active shampoo without ketoconazole, but the comparator was a pure placebo shampoo. Some of the difference might reflect mild benefit of being shampoo-active rather than ketoconazole-active.
- Single-investigator group (the Piérard team). No independent replication of this specific design in 27 years.
Despite all of that, the result is replicable in spirit (next section), the mechanism is now understood (Inui & Itami 2007), and the effect direction is consistent with everything since.
The mechanism: it’s not just dandruff treatment
Inui & Itami 2007, published as a short communication in the Journal of Dermatological Science, articulated what we now understand as the dual mechanism:
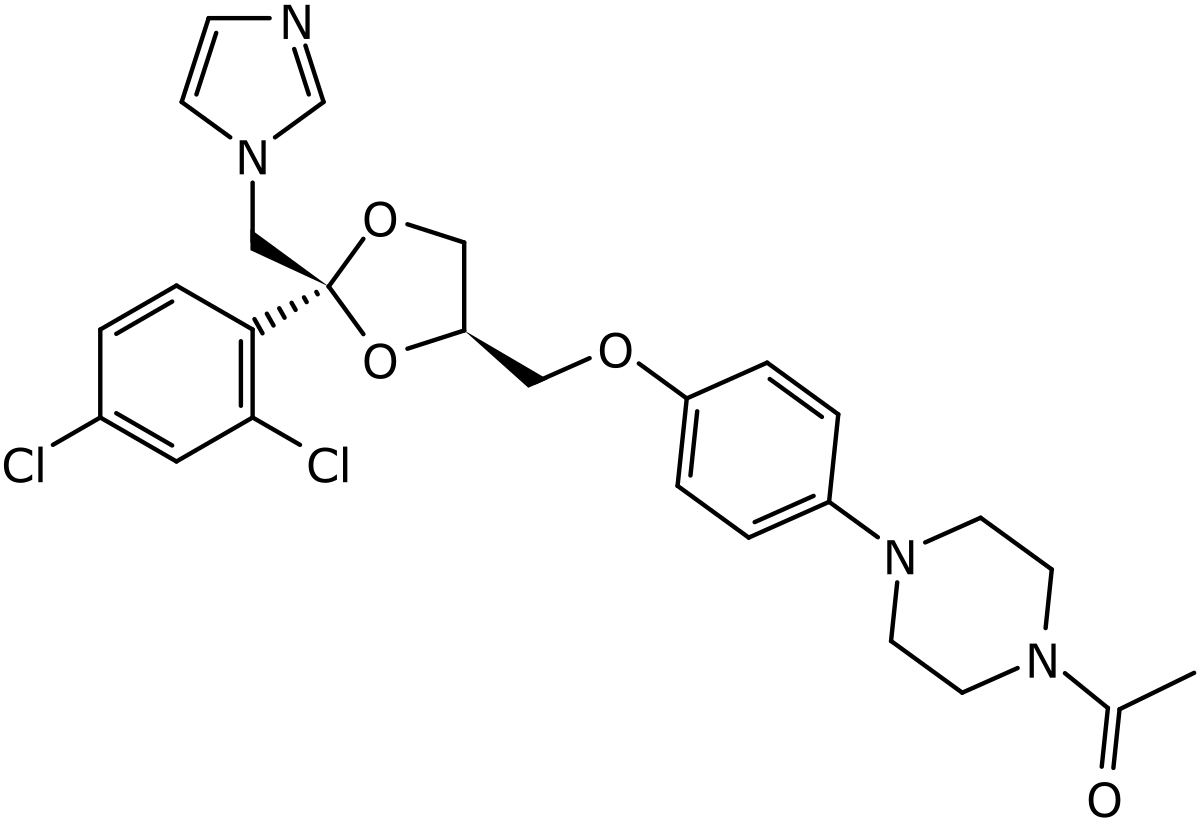
Ketoconazole structure by Mrgreen71/BartVL71 via Wikimedia Commons (CC BY-SA 3.0). The imidazole ring on the right is the antifungal pharmacophore; the molecule’s broader steroid-modulating activity comes from cytochrome P450 interaction.
Mechanism 1: anti-androgenic activity (the AGA-specific pathway)
Ketoconazole is a potent inhibitor of cytochrome P450 enzymes, including those in the steroid synthesis pathway. Specifically:
- It interferes with 5α-reductase activity at the scalp (the same enzyme finasteride targets) — though much more weakly than finasteride and only locally
- It inhibits steroidogenesis in adrenal and gonadal tissue (the basis of its old systemic use as an anti-androgen for prostate cancer)
- Topical application means the systemic anti-androgen effect is negligible, but the local scalp DHT-modulating effect is plausibly real
This is why ketoconazole shampoo benefits men without dandruff. It’s not (only) treating Malassezia — it’s also blunting local DHT signalling at the follicle.
Mechanism 2: anti-Malassezia and anti-inflammatory action
Malassezia species are the dominant yeast on human scalp. They feed on sebaceous lipids, and their metabolic products (free fatty acids, indoles) drive scalp inflammation. In AGA, follicular inflammation is a documented contributor to miniaturisation — not the main driver, but a contributing factor.
Reducing Malassezia load:
- Reduces inflammatory infiltrate around miniaturising follicles
- Improves the local follicle environment for anagen induction
- Has the side benefit of treating any subclinical dandruff
These two mechanisms explain why ketoconazole produces a measurable AGA effect even in patients without overt dandruff, and why it’s complementary to (not redundant with) finasteride and minoxidil.
The female-pattern hair loss replication: El-Garf 2019
The strongest modern data point is El-Garf et al., Biomedical Dermatology 2019 — a head-to-head trial of 2% topical ketoconazole vs 2% minoxidil in women with FPHL.
- n = 40 women (20 per arm), randomised
- Group A: 2% topical minoxidil solution
- Group B: 2% topical ketoconazole preparation
- Duration: 6 months
- Outcomes: clinical assessment + trichoscopic evaluation
Results:
| Outcome | Minoxidil 2% (Group A) | Ketoconazole 2% (Group B) |
|---|---|---|
| Significant improvement at month 4 | Yes | No |
| Significant improvement at month 6 | Yes | Yes |
| Side effects | Higher rate (typical for minoxidil scalp irritation) | 10% (mild, transient) |
| Patient satisfaction | Comparable to ketoconazole | Comparable to minoxidil |
Honest interpretation:
- Both treatments worked at 6 months. That’s two trials now confirming ketoconazole’s effect (1998 in men, 2019 in women).
- Minoxidil was faster acting. If you’re impatient or have a more aggressive miniaturisation pattern, minoxidil’s 4-month onset is meaningful.
- Ketoconazole had a much better tolerability profile — only 10% reported any side effect, compared to the 20-40% scalp irritation rates typical for minoxidil in other trials.
- Patient satisfaction didn’t differ — so the user-experience trade-off (slower but gentler) lands roughly at par.
This trial is the strongest single piece of evidence that ketoconazole is doing real work on AGA, not just treating an underlying dermatitis. Two RCTs, 27 yrs
What the systematic review concluded
Fields, Vonu, Monir, and Schoch published Topical ketoconazole for the treatment of androgenetic alopecia: A systematic review in Dermatologic Therapy (2020). Their methodology and findings:
- 47 papers screened → 9 eligible → 7 included
- 2 animal studies (n=40 mice/rodents)
- 5 human studies (n=318 patients total)
- Outcomes synthesised: hair shaft diameter, density, anagen ratio, satisfaction
Their bottom line, verbatim: “Topical ketoconazole is a promising adjunctive or alternative therapy in the treatment of AGA, though randomized controlled trials are needed.”
That’s a calibrated conclusion, not an endorsement. It says: the data are positive but the methodological rigour isn’t yet at the level we’d require for a definitive guideline statement. Most of the included trials had small samples, weren’t fully blinded, or had design quirks.
What this means in practice: if a user with AGA tells you they want to try ketoconazole shampoo, the evidence base supports that decision. It does not yet support naming ketoconazole as a first-line or even officially second-line treatment in dermatology guidelines.
Position in a 2026 stack
Where does ketoconazole shampoo realistically fit relative to other tools?
| Treatment | Evidence weight | Cost/month | Daily time | When to use |
|---|---|---|---|---|
| Topical minoxidil 5% | RCT-grade, decades | $20-50 | 5-10 min × 2/day | First-line topical |
| Oral finasteride 1mg | RCT-grade, decades | $5-15 | seconds (oral) | First-line oral |
| Microneedling 1.5mm | Multiple RCTs (Dhurat 2013, 2025 meta) | $15 (one-time) | 10 min × weekly | Strong adjunct |
| Ketoconazole 2% shampoo | Multiple supportive trials, 1998–2019 | $10-25 | 0 added (replaces shampoo) | Easy add-on |
| Saw palmetto 320mg/day | One major RCT, weak meta-analyses | $15-25 | seconds (oral) | Natural DHT alt |
| Tocotrienols 100mg/day | One RCT (Beoy 2010) | $15-25 | seconds (oral) | Antioxidant axis |
| GHK-Cu peptide | One indirect trial, weak | $30-90 | 2 min × daily | Experimental |
Key insight: ketoconazole is the only entry on this list with zero added time cost — you were going to shampoo anyway. That alone makes it one of the most underused interventions for users who are otherwise complaining about how time-consuming their stack is.
Practical use protocol
Concentration: 2% prescription Nizoral if available (used in trials); 1% OTC (Nizoral A-D in the US) is the practical default. The 1%-vs-2% gap has not been formally tested for AGA — most users get away with 1%.
Frequency: 2-4× per week. Daily use can dry out the scalp.
Application:
- Wet hair fully
- Apply enough shampoo to lather across the entire scalp (not just where you see thinning)
- Lather thoroughly
- Leave for 3-5 minutes before rinsing — this is critical. Rinsing immediately wastes 80% of the active ingredient’s contact time with the scalp.
- Rinse fully
- On non-ketoconazole days, use a normal gentle shampoo
Combining with minoxidil: shampoo on a ketoconazole day, dry your scalp completely, then apply minoxidil. The order matters because ketoconazole wants direct scalp contact, and minoxidil needs a dry application surface for absorption.
Combining with microneedling: don’t apply ketoconazole shampoo within 24 hours of microneedling — the open microchannels increase absorption and irritation risk unpredictably.
Cost reality
| Product | Strength | Bottle price | Daily cost (3×/week) |
|---|---|---|---|
| Nizoral A-D OTC (US) | 1% | ~$15-18 / 7 oz | ~$0.30 |
| Nizoral 2% (prescription, US/UK) | 2% | ~$25-40 / 4 oz | ~$0.80 |
| Generic ketoconazole 1% (Amazon, brand-equivalent) | 1% | ~$10-15 / 7 oz | ~$0.25 |
| Generic ketoconazole 2% (international, mail-order) | 2% | ~$15-25 / 7 oz | ~$0.40 |
| Compound pharmacy 2% | 2% | $30-50 / 200ml | ~$0.50 |
For comparison, GHK-Cu serums typically run $30-90/month with much weaker evidence. Ketoconazole is the value play of the OTC topical world.
What ketoconazole won’t fix on its own
Honest expectation-setting:
- Aggressive Norwood IV-V miniaturisation. Ketoconazole’s effect size is modest; you need finasteride or dutasteride for the underlying DHT axis.
- Fast-progressing AGA in your 20s. This is a “use it but don’t rely on it” situation. Add minoxidil + DHT blocker as the primary stack.
- Telogen effluvium with a clear trigger. Iron deficiency, thyroid dysfunction, severe stress, post-COVID, postpartum — these need addressing first. Ketoconazole is irrelevant.
- Scarring alopecias (lichen planopilaris, frontal fibrosing alopecia, CCCA). Different pathology. Ketoconazole is unhelpful and may irritate already-inflamed scalp.
- Alopecia areata. Autoimmune, not androgenic. Different disease, different treatment.
Decision card
Use ketoconazole shampoo if any of these is true:
- You’re starting any AGA stack — it’s a free add-on with real evidence
- You’re already on minoxidil + finasteride and want to add one more low-cost shot on goal
- You have any sign of seborrheic dermatitis or dandruff alongside AGA (you’ll get both effects)
- Cost or time-budget is a constraint and you want maximum evidence per dollar
Skip ketoconazole if any of these is true:
- You expect dramatic regrowth from shampoo alone — not realistic
- You’re allergic to imidazole antifungals
- You have aggressive scalp inflammation that isn’t seborrheic — see a dermatologist first
- You can’t commit to 6+ months of consistent use — early evaluation will frustrate you
Re-evaluate at 6 months. Take photos at baseline. The Piérard-Franchimont data suggest density change emerges around month 6 — earlier evaluation is a waste of judgment.
Related reading
- Minoxidil complete guide — the foundation underneath any modern AGA stack
- DHT and hair loss — why ketoconazole’s anti-androgenic effect matters
- Finasteride safety data — the systemic DHT-blocking option
- Microneedling guide — the strongest adjunct for users willing to add weekly time
- GHK-Cu copper peptide — for contrast: how a topical with weak evidence compares
- Best hair loss treatments 2026 — top-of-funnel decision tree
References
- Piérard-Franchimont C, De Doncker P, Cauwenbergh G, Piérard GE. Ketoconazole shampoo: effect of long-term use in androgenic alopecia. Dermatology 1998;196(4):474-477. PMID 9669136.
- Inui S, Itami S. Reversal of androgenetic alopecia by topical ketoconazole: relevance of anti-androgenic activity. Journal of Dermatological Science 2007;45(1):66-68. PMID 16997533.
- El-Garf A, Ahmed E, Hassan S. Trichogenic effect of topical ketoconazole versus minoxidil 2% in female pattern hair loss: a clinical and trichoscopic evaluation. Biomedical Dermatology 2019;3(1):8.
- Fields JR, Vonu PM, Monir RL, Schoch JJ. Topical ketoconazole for the treatment of androgenetic alopecia: A systematic review. Dermatologic Therapy 2020;33(1):e13202. PMID 31858672.
- Hugo Pérez BS. Ketoconazole as an adjunct to finasteride in the treatment of androgenetic alopecia in men. Medical Hypotheses 2004;62(1):112-115.
Published May 2026. Topical ketoconazole 1% is OTC in most countries; 2% requires prescription in some jurisdictions. Side effects are typically mild and transient — patch-test if you have sensitive skin or known azole allergy. Topical use does not carry the systemic hepatotoxicity warnings associated with oral ketoconazole. For severe AGA, this article does not replace consultation with a board-certified dermatologist.